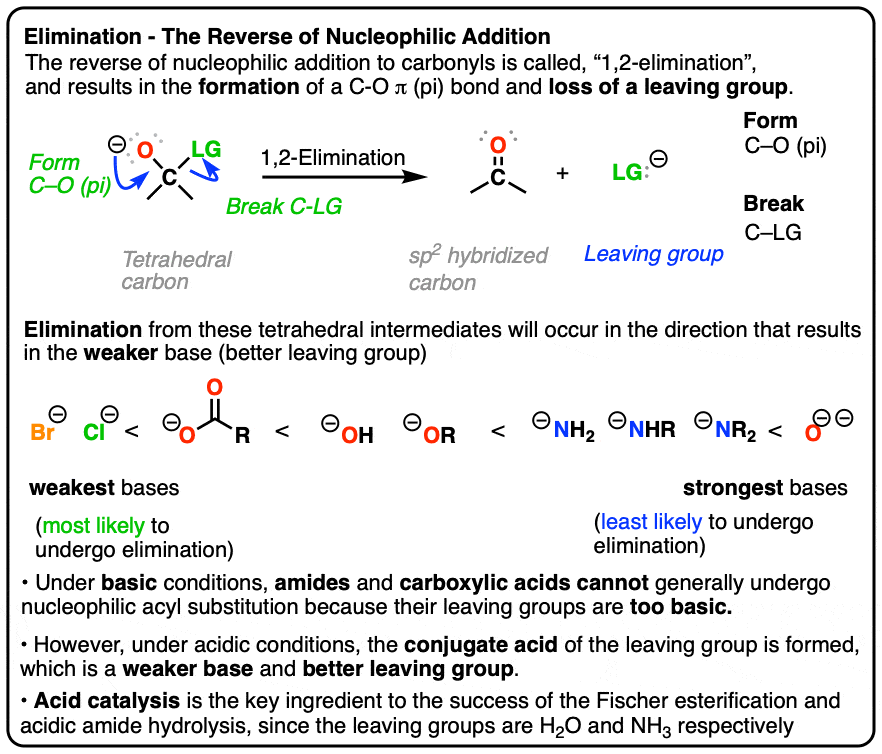
Calcium carbonate reacts with aqueous HCl to give CaCl2 and CO2 according to the reaction, CaCO3 (s) + 2HCl (aq) →CaCl2 (aq) + CO2 (g) + H2O (l) .What mass of CaCO3
SOLVED: Calcium carbonate is added to separate solutions of hydrochloric acid and ethanoic acid of the same concentration. State one similarity and one difference in the observations you could make. (i) Write

Advanced Amino Acid-Based Technologies for CO2 Capture: A Review | Industrial & Engineering Chemistry Research

Acids and Metals Acids react with certain metals to produce hydrogen gas and the metal salt. metal acid metal salt. - ppt download

Write symbolic representation for the word equations and balance Sodium carbonate + Hydrochloric acid (dil) to Sodium chloride + Water + Carbon dioxide

Law of Conservation Mass reacting masses equations demonstration experiment gcse chemistry Calculations gcse chemistry igcse KS4 science A level GCE AS A2 O Level practice questions exercises