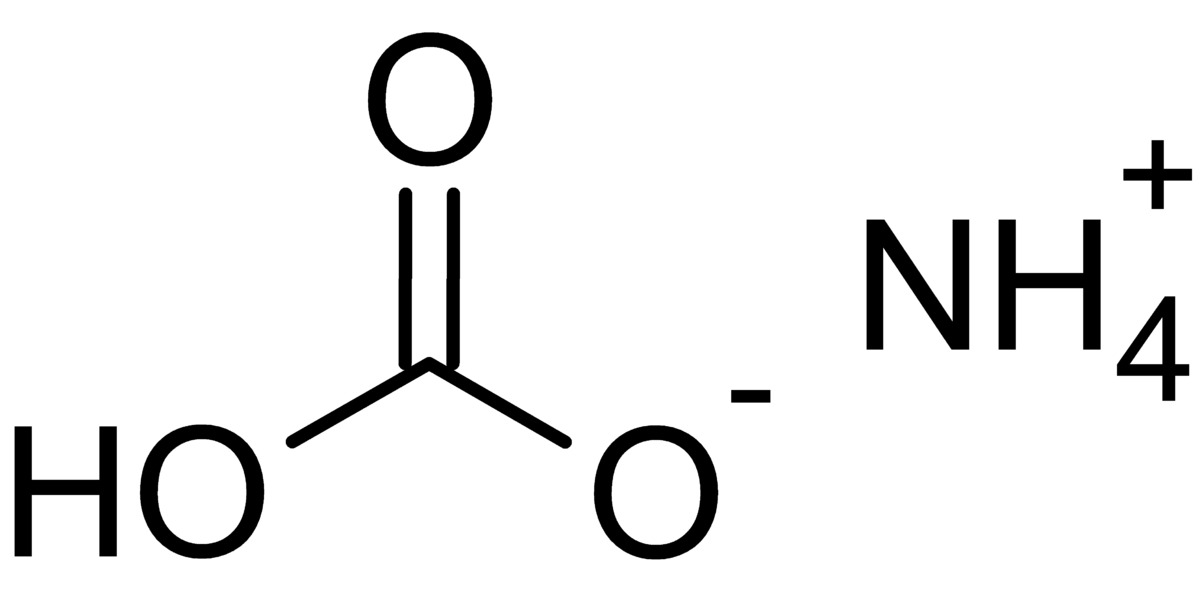
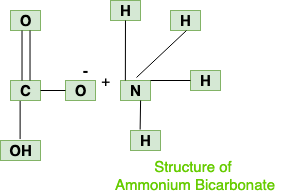
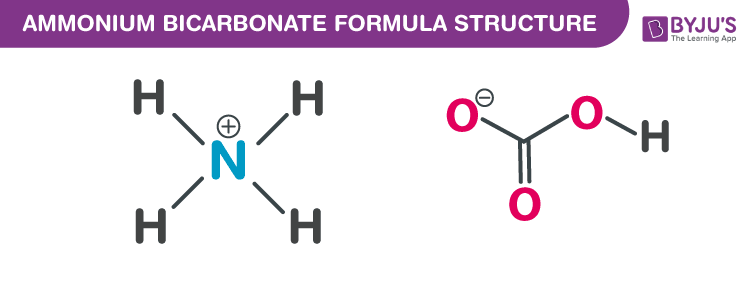
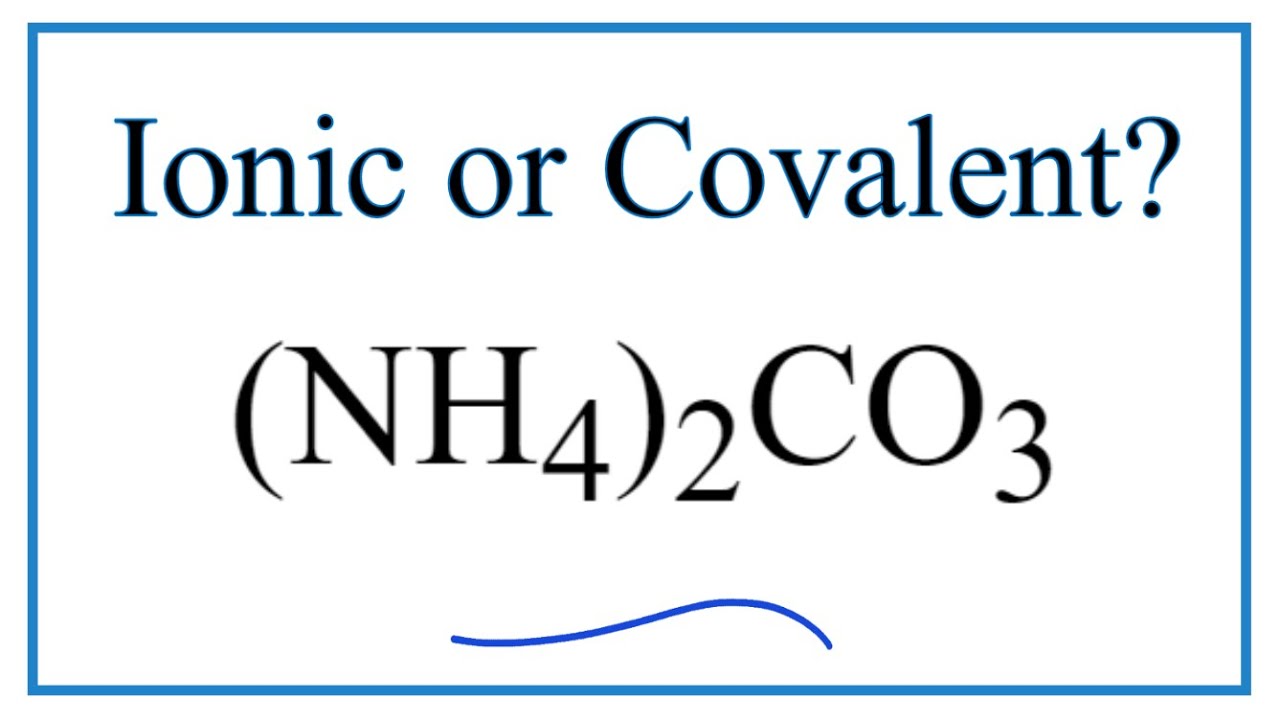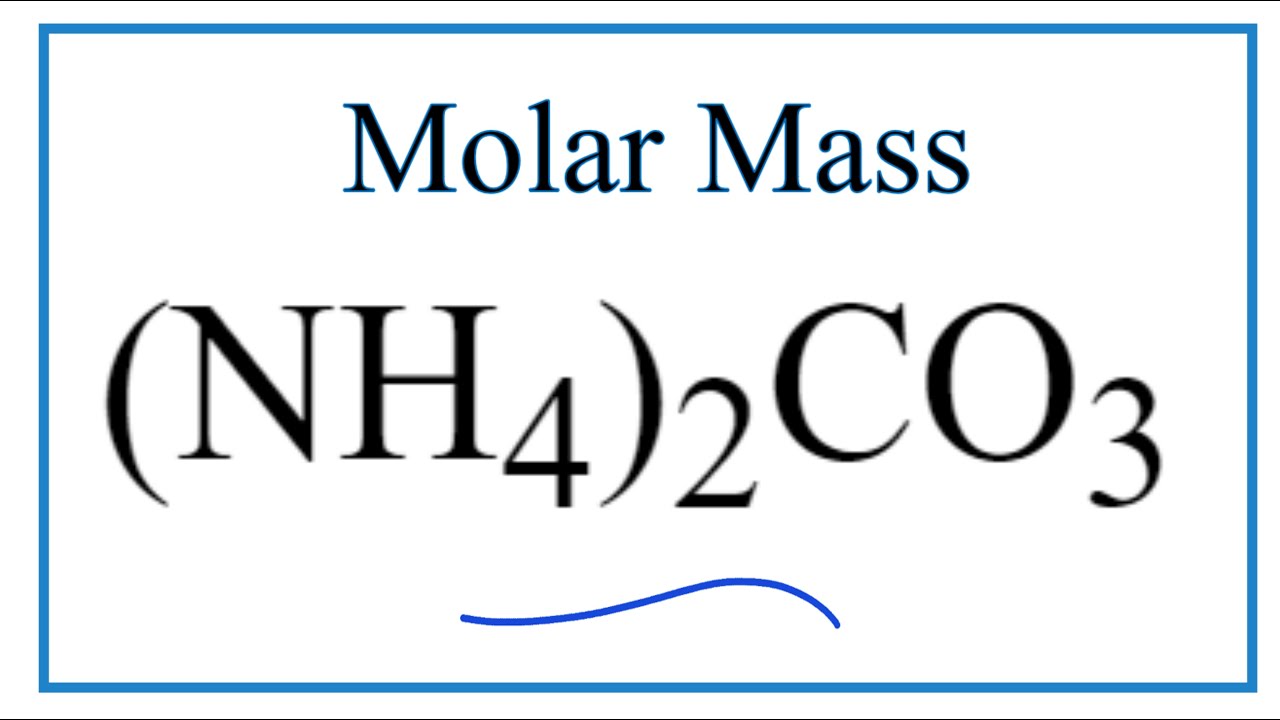Naming and writing formulas. Ammonium NH 4 1+ Calcium Ca 2+ Iron (II) Fe 2+ Iron (III) Fe 3+ Sodium Na 1+ Tin (IV) Sn 4+ Acetate C 2 H 3 O 2 1- Carbonate. - ppt download

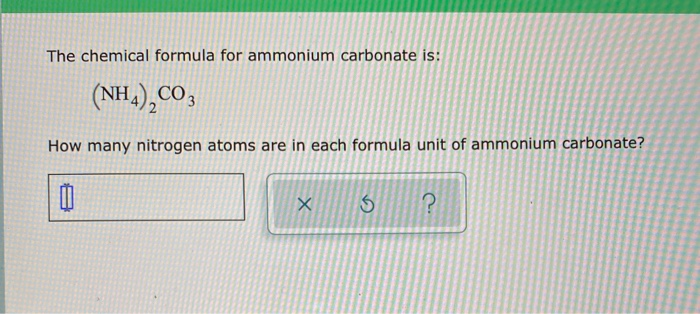
What is the formula of the compound formed between the ammonium ion and the carbonate ion? A. NH4CO3 B. NH4(CO3)2 C. (NH4)2CO3 D. (NH4)3CO3 | Homework.Study.com
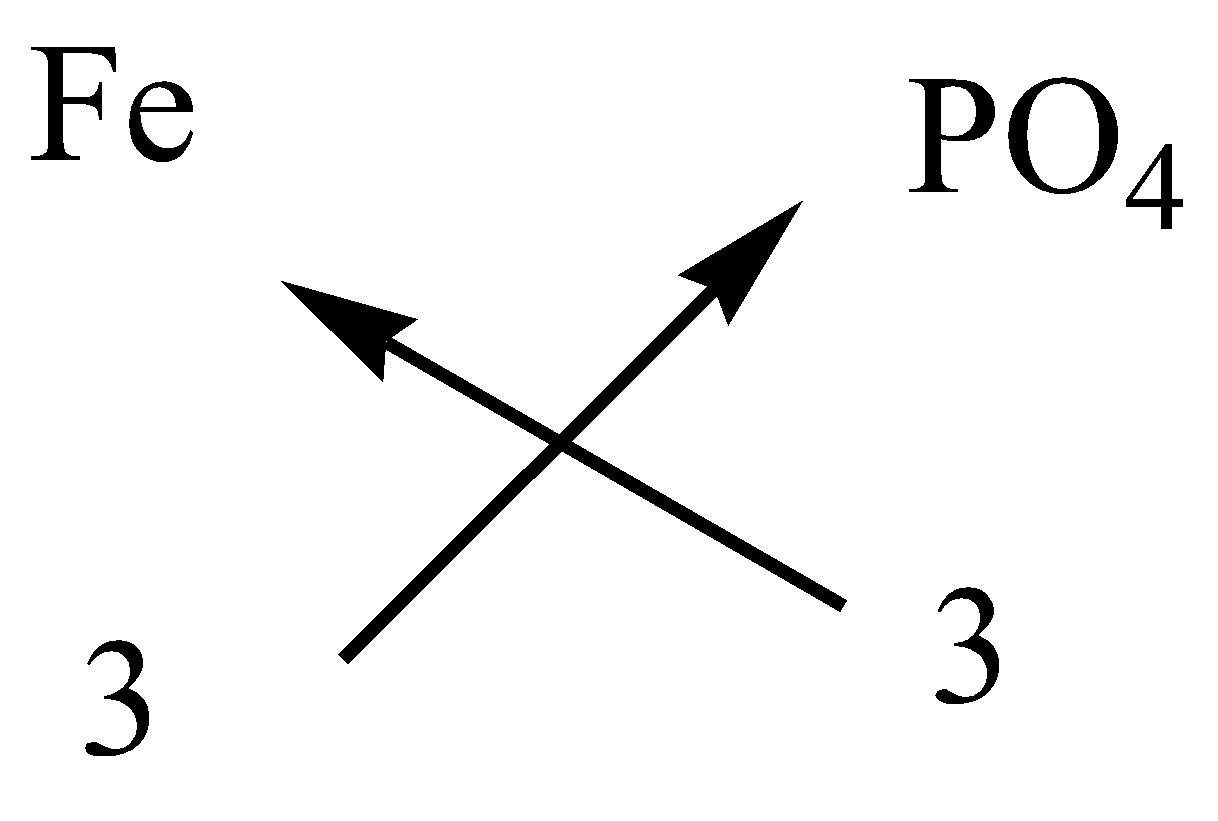
Write the formula of the following compounds by criss cross methodA.Ammonium carbonateB.Calcium bicarbonateC.Ferric phosphateD.Potassium sulfateE.Sodium zincate

Negative ion Formula in compoundcharge Oxide Hydroxide Nitrate NO 3 -1 Sulphate Carbonate. - ppt download
![If the relative molecular mass of ammonium nitrate is 80, the percentage of nitrogen and oxygen in ammonium nitrate is: [N = 14, H = 1, O = 16] If the relative molecular mass of ammonium nitrate is 80, the percentage of nitrogen and oxygen in ammonium nitrate is: [N = 14, H = 1, O = 16]](https://dwes9vv9u0550.cloudfront.net/images/3796715/307077b2-1bb0-4bde-a9d0-fe5f6659fec9.jpg)
If the relative molecular mass of ammonium nitrate is 80, the percentage of nitrogen and oxygen in ammonium nitrate is: [N = 14, H = 1, O = 16]

Chemical formula : 1) ammonium carbonate - Science - Atoms and Molecules - 11046099 | Meritnation.com

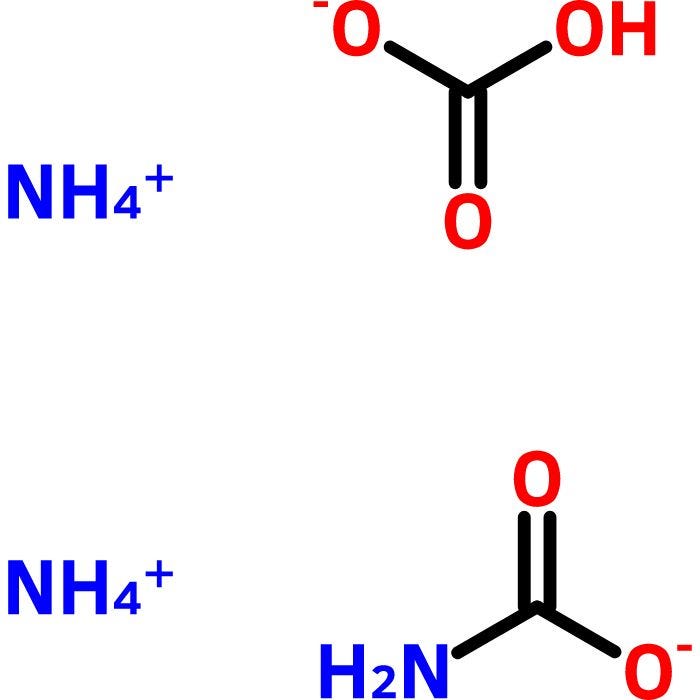
![Ammonium Carbonate [(NH4)2CO3] Molecular Weight Calculation - Laboratory Notes Ammonium Carbonate [(NH4)2CO3] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/03/ammonium-carbonate-molecular-weight-calculation-300x198.jpg)