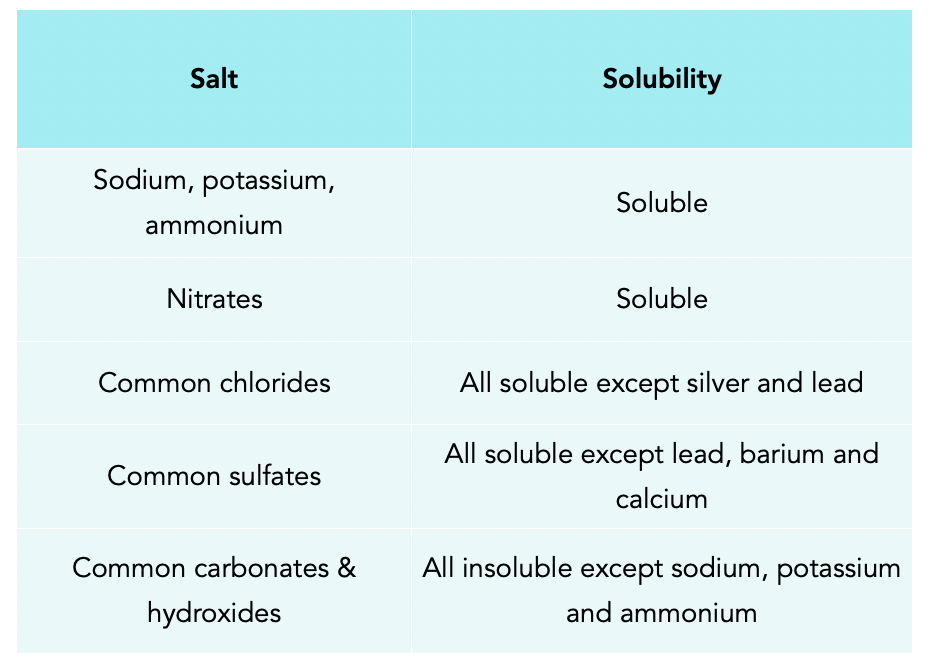
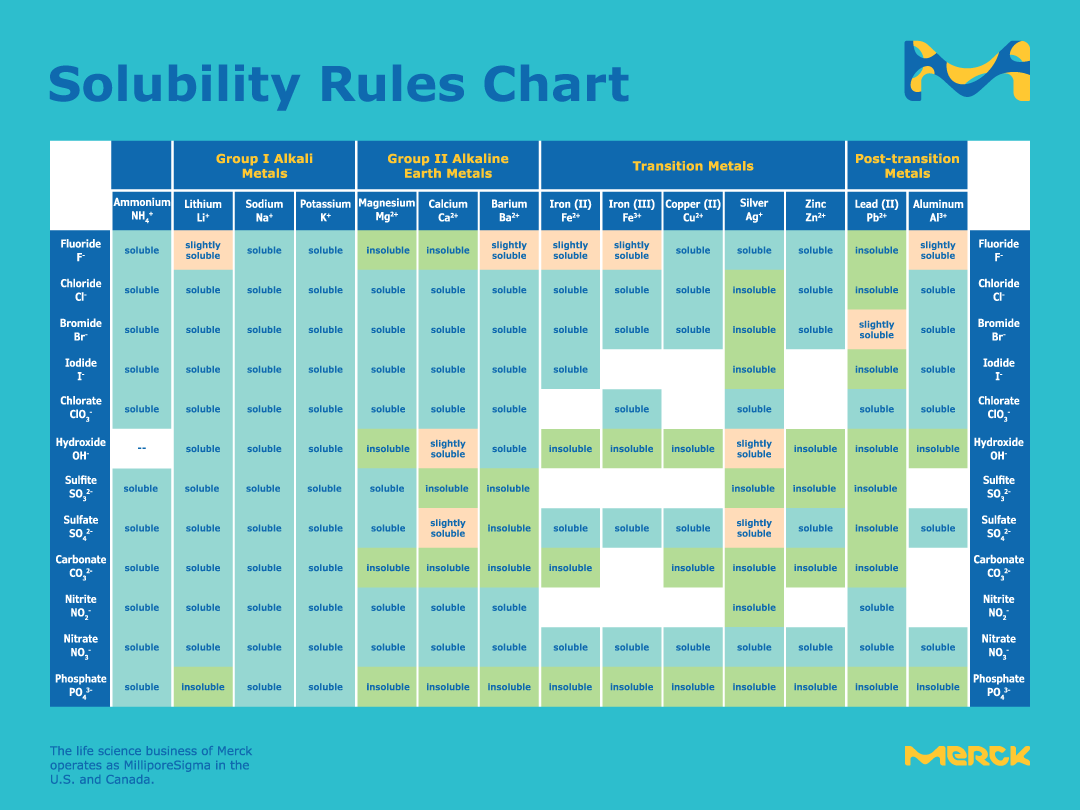
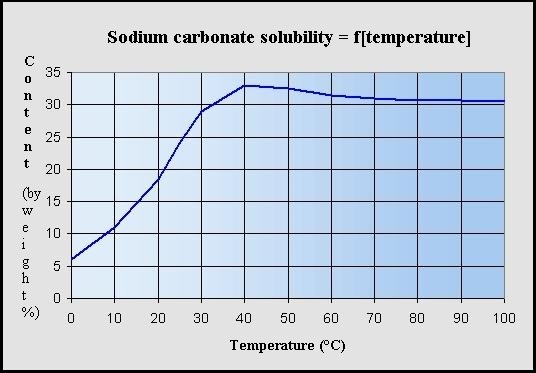
What is the trend in solubility of carbonates, bicarbonates, nitrates, hydroxides of alkali and alkaline earth metals

Table 14 from IUPAC-NIST Solubility Data Series. 95. Alkaline Earth Carbonates in Aqueous Systems. Part 1. Introduction, Be and Mg | Semantic Scholar

Why does the solubility of alkaline earth metal carbonates and sulphates in water decrease down the group ?
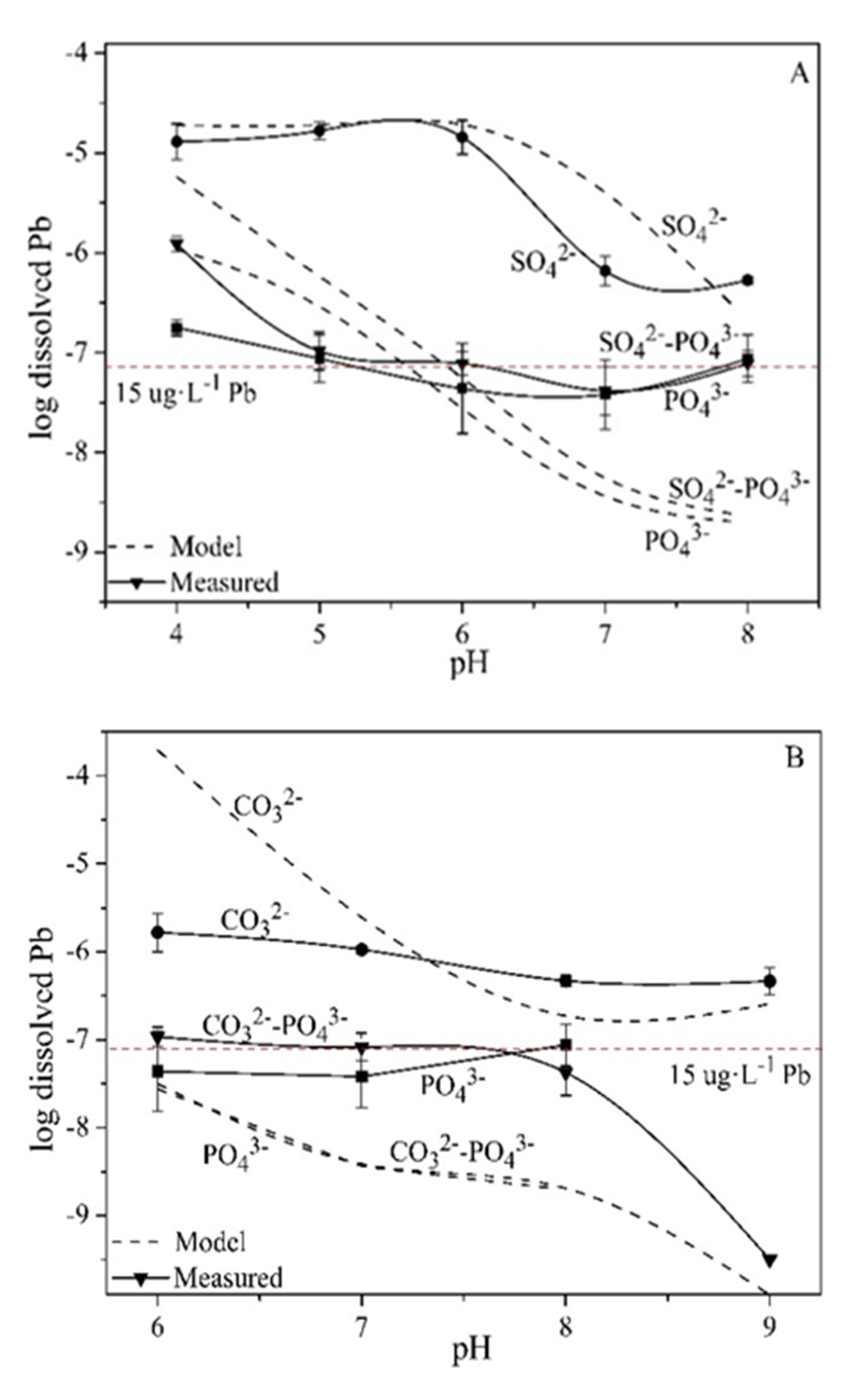
Minerals | Free Full-Text | Pb Mineral Precipitation in Solutions of Sulfate, Carbonate and Phosphate: Measured and Modeled Pb Solubility and Pb2+ Activity

inorganic chemistry - Why exception is noted in the solubility of lead salts? - Chemistry Stack Exchange