BCC - Blind Carbon Copy Acronym, Technology Concept Background Stock Illustration - Illustration of copy, blog: 196334889
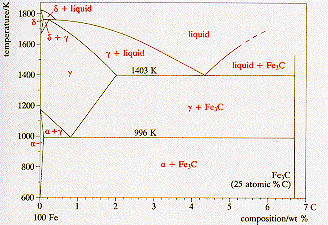
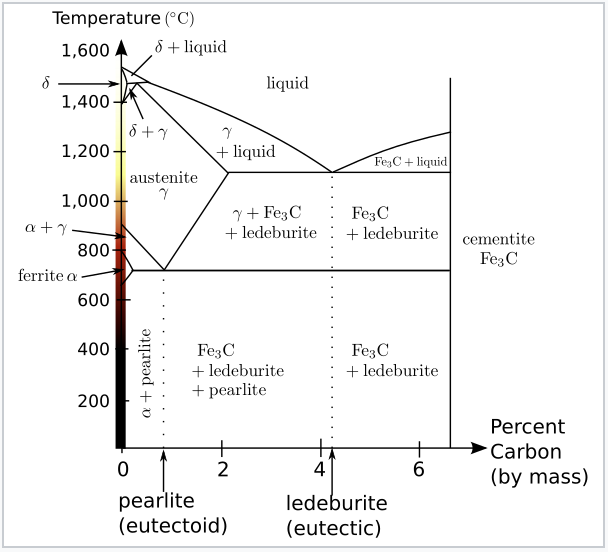
Tetragonality of bcc-Fe as a function of carbon content. Solid marks... | Download Scientific Diagram
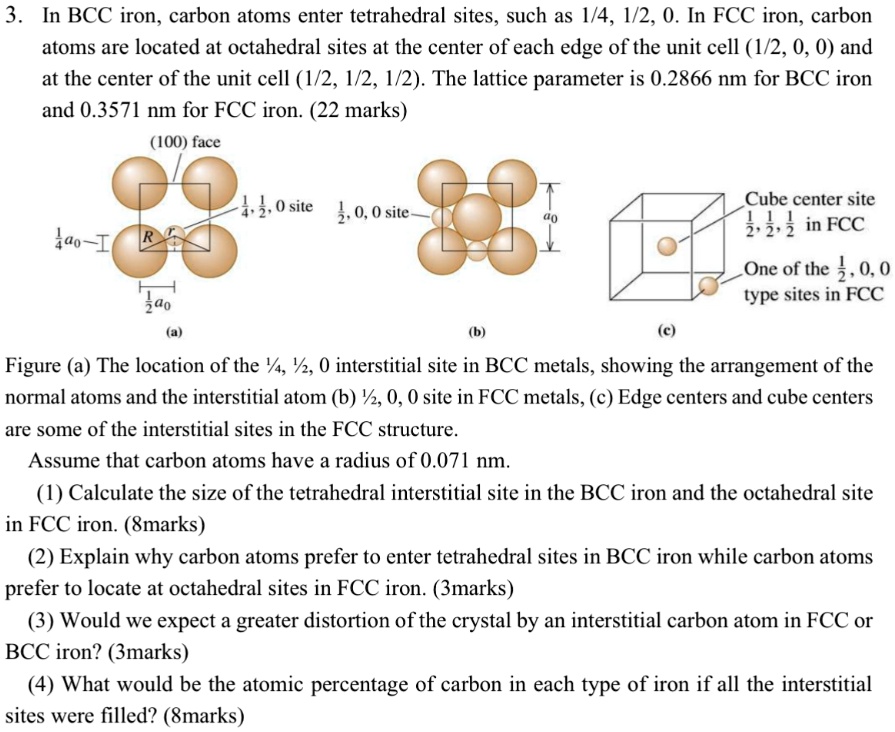
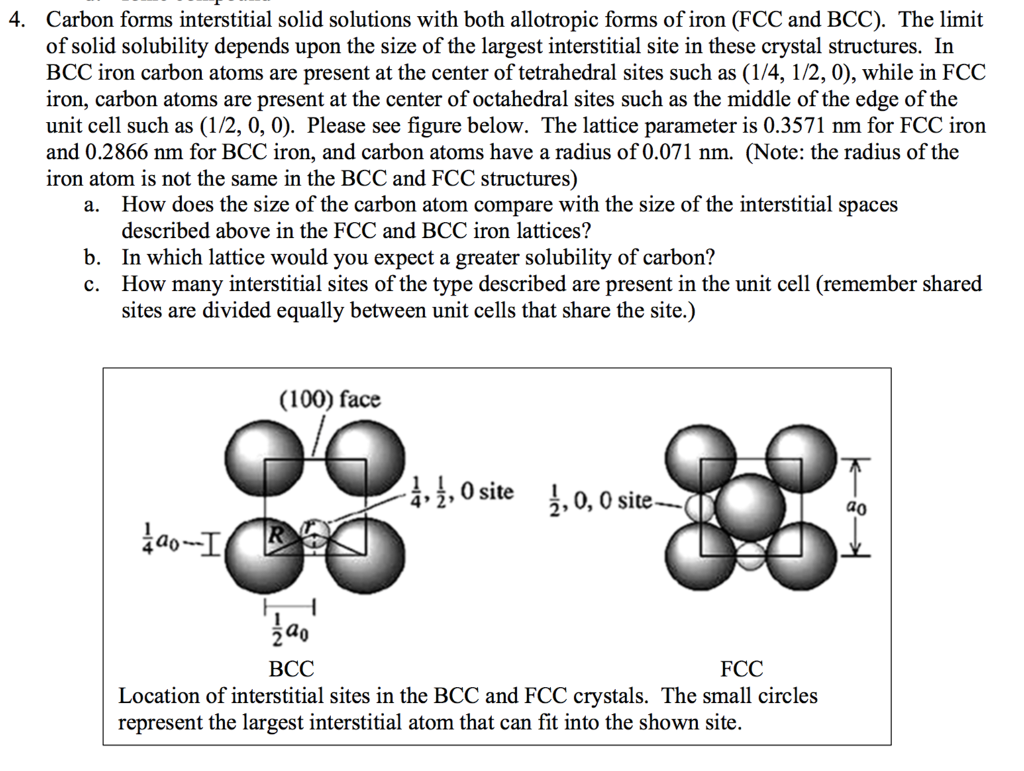
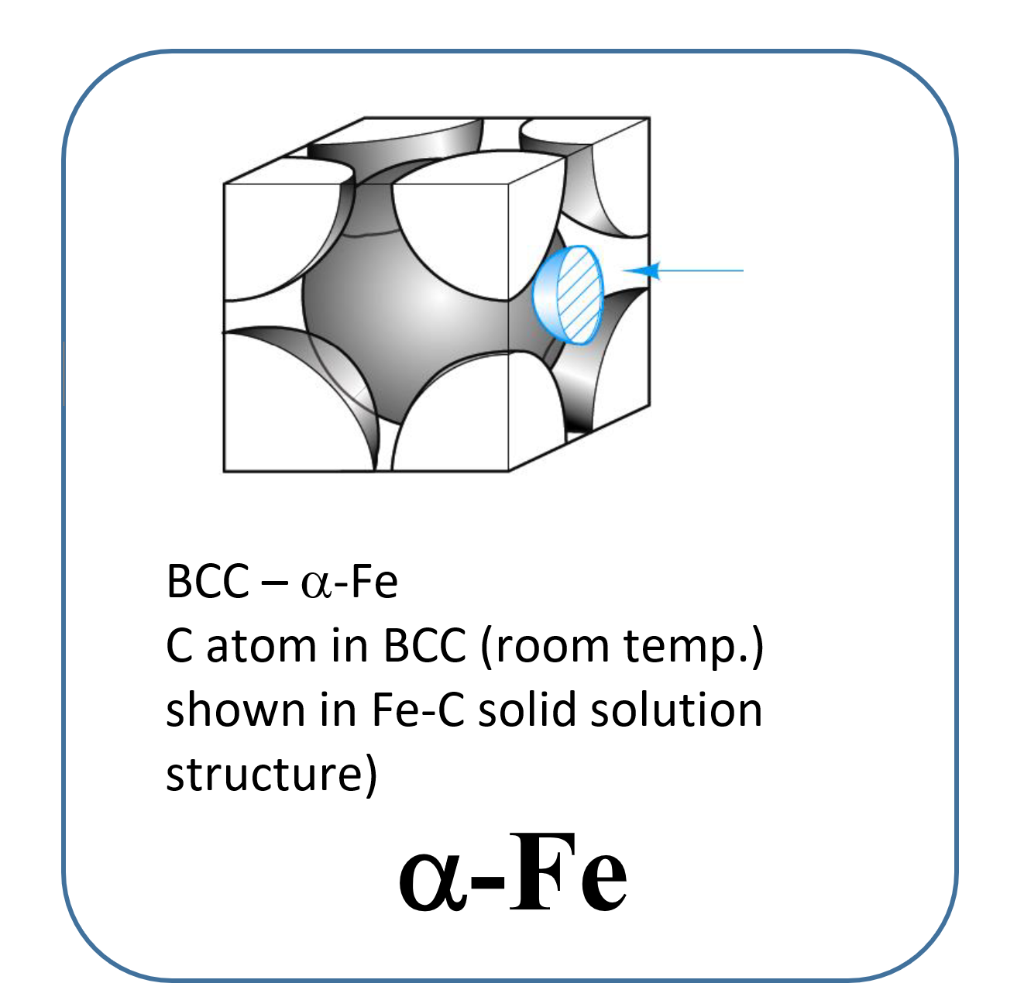
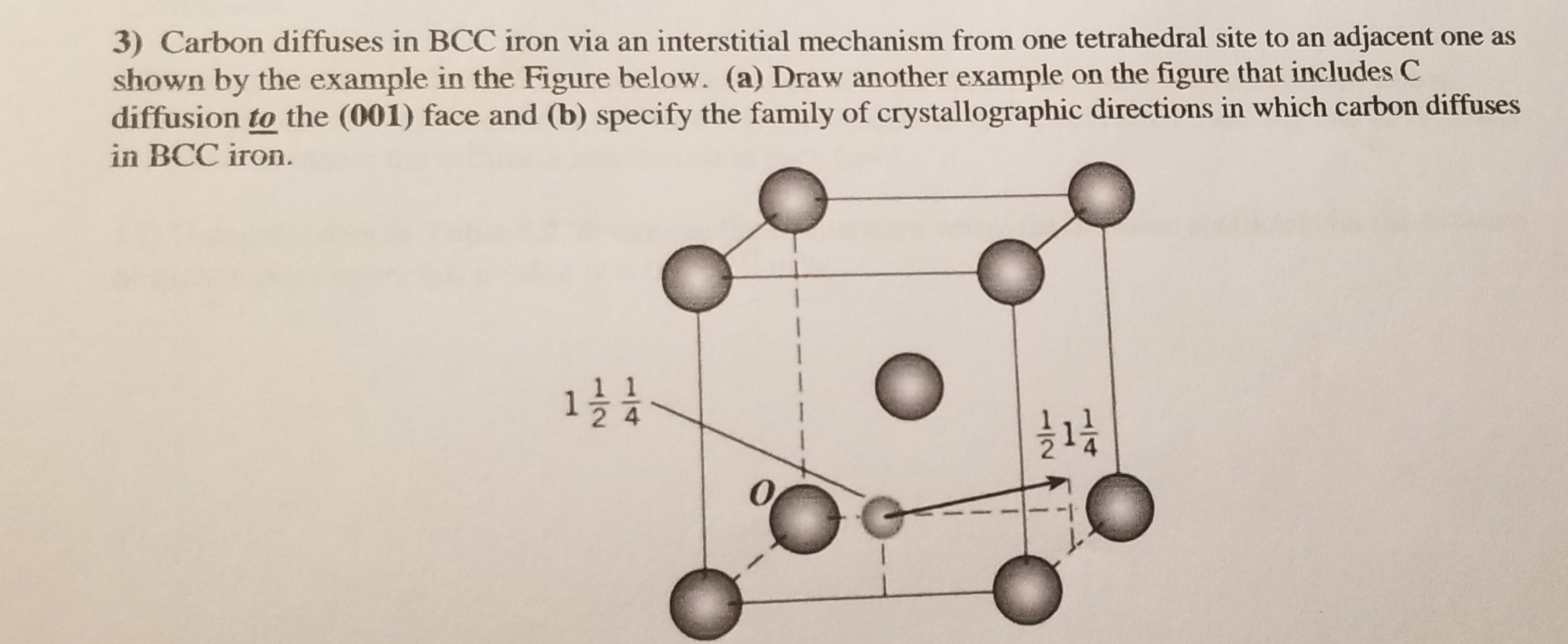
SOLVED: 3 In BCC iron, carbon atoms enter tetrahedral sites, such as 1/4, 1/2, 0. In FCC iron, carbon atoms are located at octahedral sites at the center of each edge of
What Is the Difference Between FCC and BCC? (Crystal Structure, Properties, Interstitial Sites, and Examples) – Materials Science & Engineering
Using the Blind Carbon Copy (BCC) Feature in Email | Information Technology | University of Pittsburgh
Using the Blind Carbon Copy (BCC) Feature in Email | Information Technology | University of Pittsburgh





:max_bytes(150000):strip_icc()/what-is-bcc-blind-carbon-copy-1171131_final_AC-5c48a51ac9e77c0001142877.png)