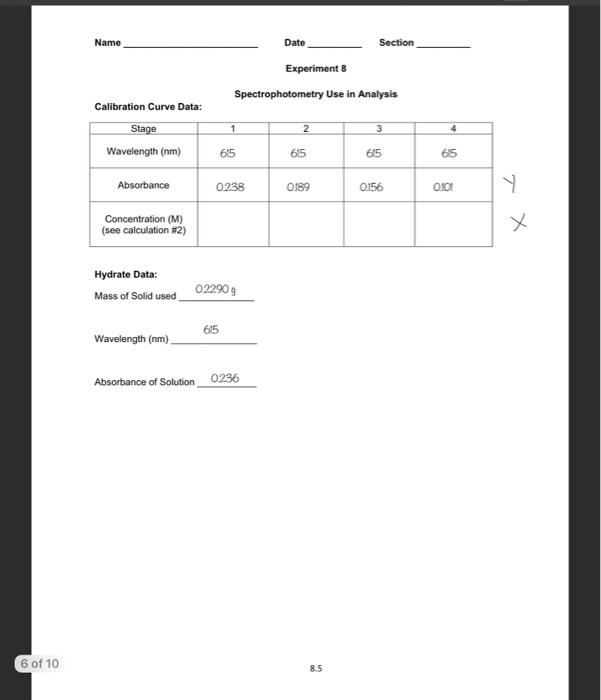
SOLVED: Use the equation C1V1 C2V2 to calculate V1 and the volume of diluent (Vd) for the following dilutions: The final volume for these dilutions is 10 mL 1/10 V1: b. 1/20

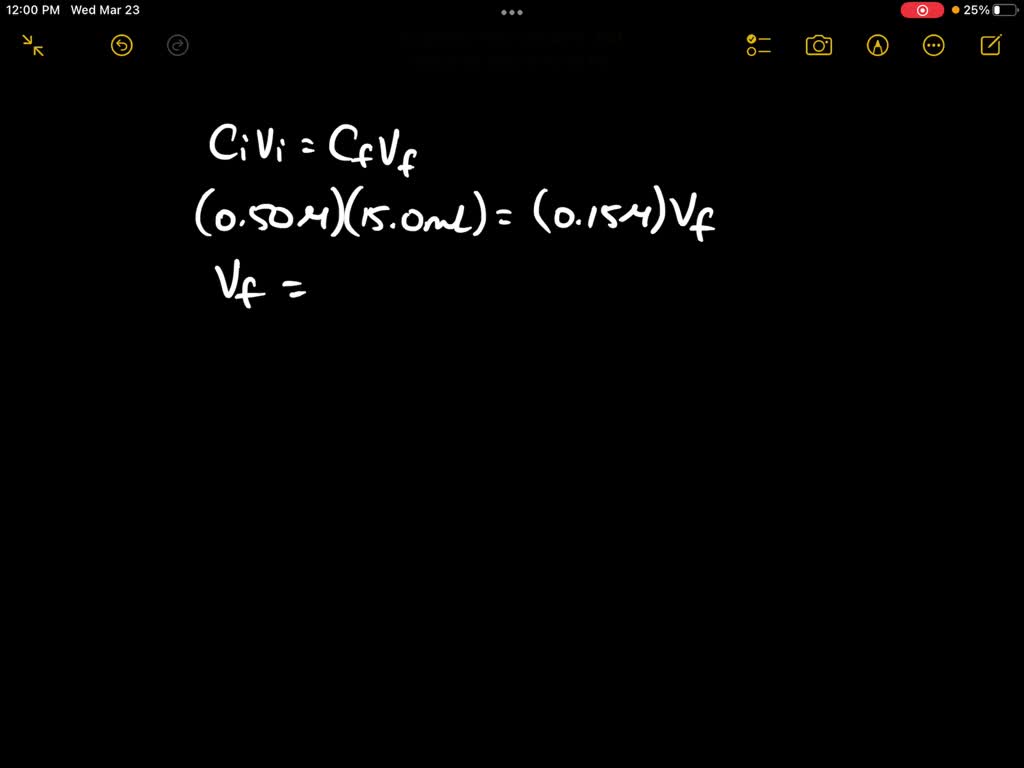
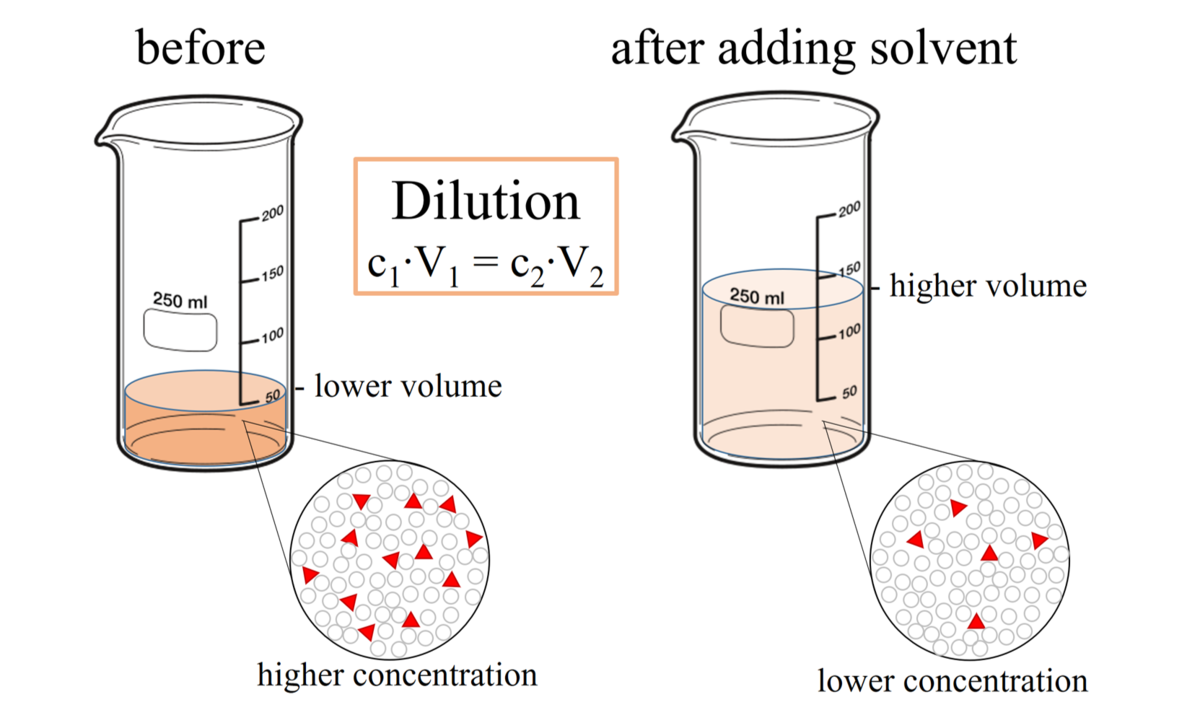
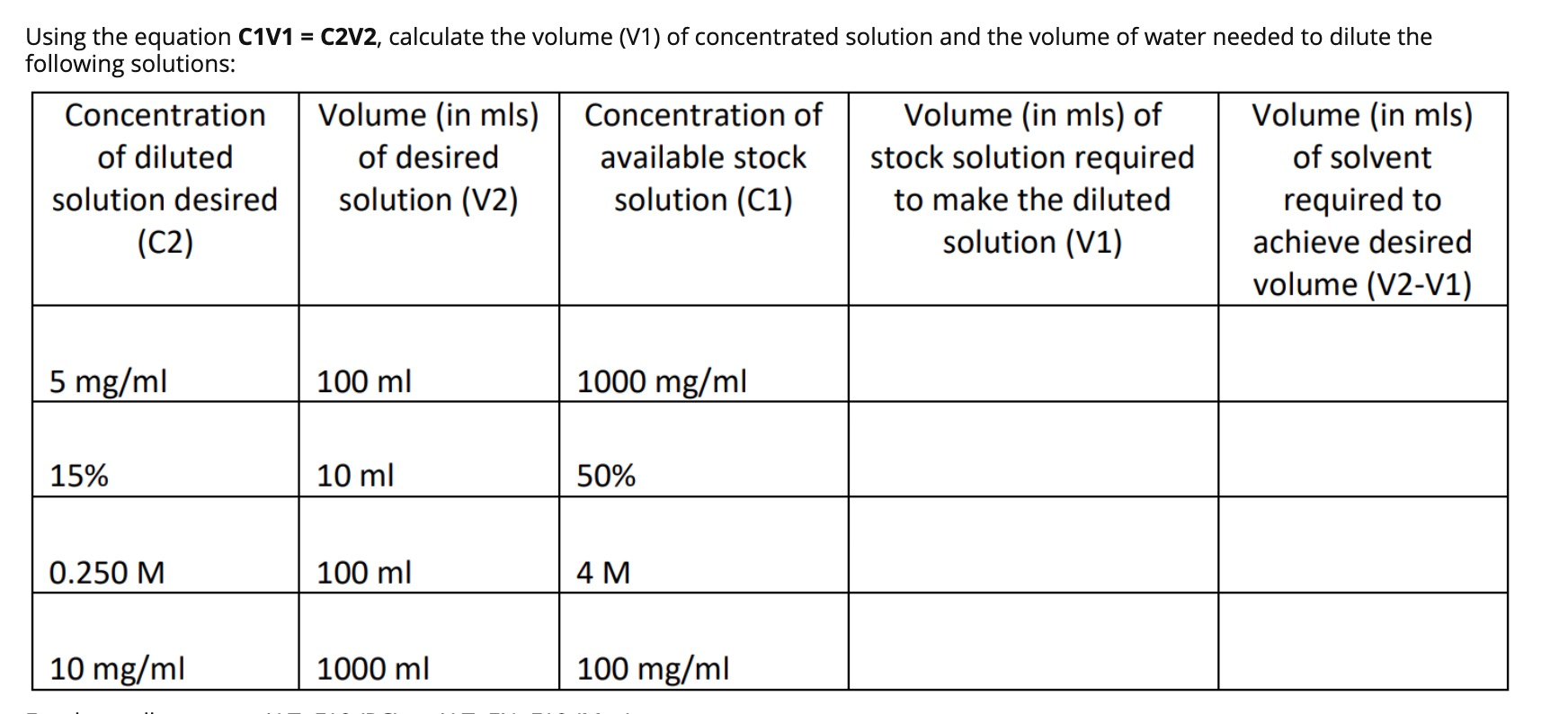
SOLVED: 1) Prepared 100 mL buffer by using 35 mL 0.2 M sodium acetate and 65 mL 0.2 M acetic acid 2) Use C1V1 = C2V2 formula to calculate final concentration of












.PNG)