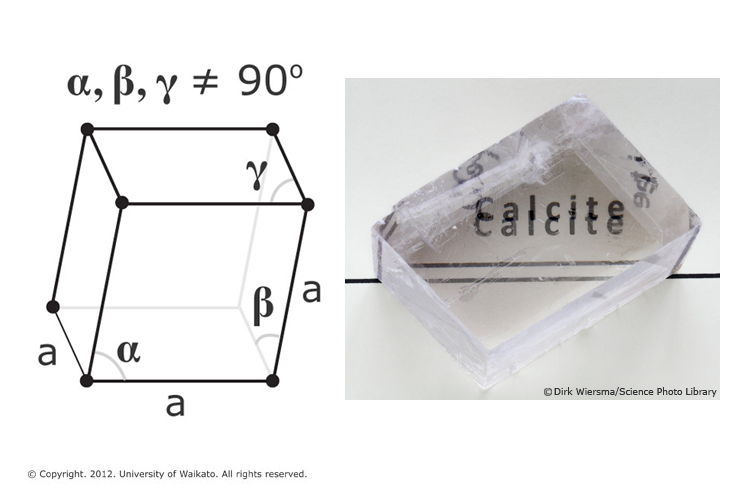
Citric Acid ,calcium Carbonate, Reacts with Water Stock Footage - Video of healthcare, single: 64804112

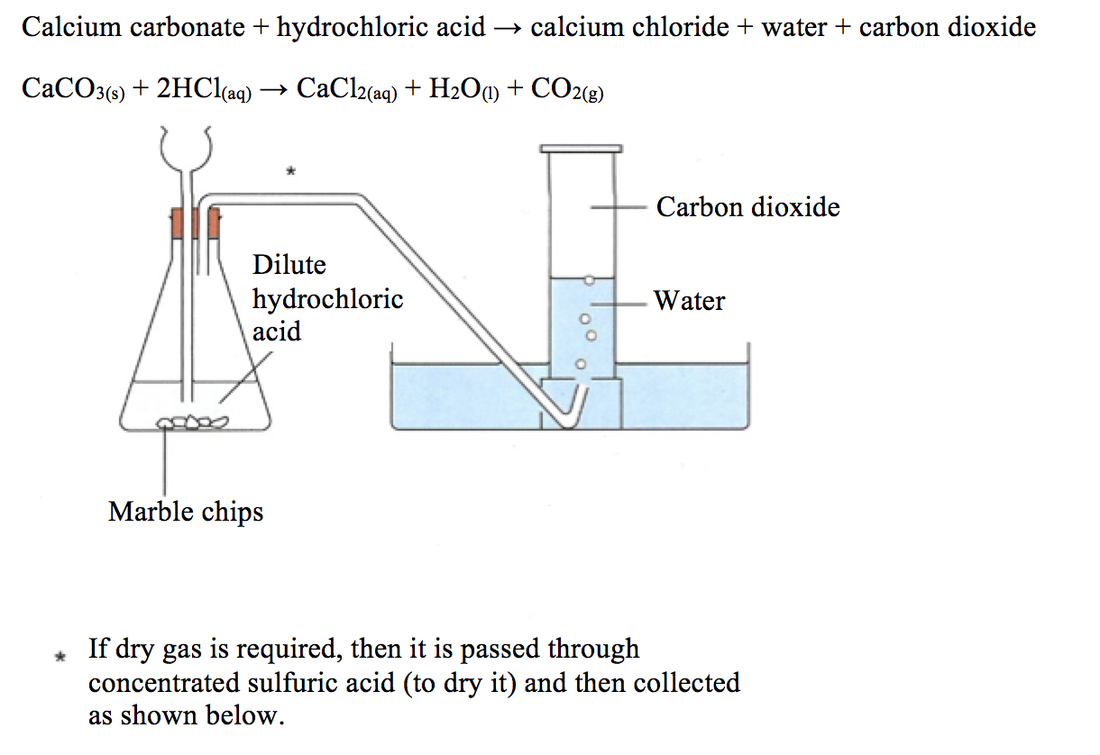
Write a balanced chemical equation for the reaction of calcium carbonate and dil. hydrochloric acid.

In an experiment, hydrochloric acid was added to calcium carbonate. These are the equations I came up with. Can somebody check them for me? Additionally, I'm thinking the net ionic equation is

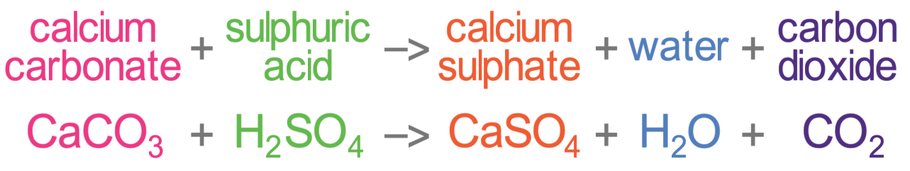
CaCO3 + 2HCl → CaCl2 + H2O + CO2 The mass of calcium chloride formed when 2.5 g of calcium carbonate is dissolved in excess of hydrochloric acid is:
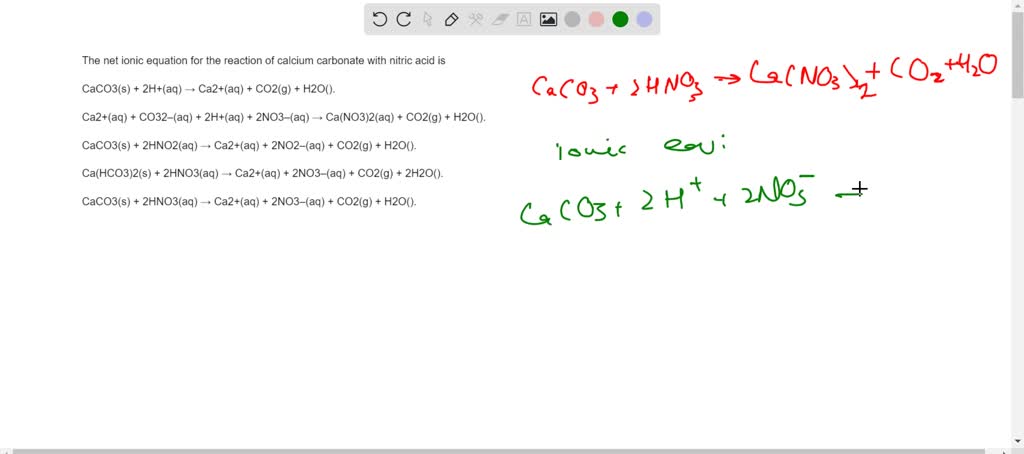
SOLVED: The net ionic equation for the reaction of calcium carbonate with nitric acid is CaCO3(s) + 2H+(aq) → Ca2+(aq) + CO2(g) + H2O(). Ca2+(aq) + CO32–(aq) + 2H+(aq) + 2NO3–(aq) →

calcium carbonate reacts with dilute hydrochloric acid to produce carbon dioxide Stock Photo - Alamy

The effect of changing the concentration of hydrochloric acid on the rate of reaction with calcium carbonate - IG Exams










![MCQ] - Which correctly represents Parent acid and base of Calcium MCQ] - Which correctly represents Parent acid and base of Calcium](https://d1avenlh0i1xmr.cloudfront.net/0014c0c3-c848-4073-a814-6e71c0e2bf5e/reaction-to-form-calcium-carbonate---teachoo-01.jpg)