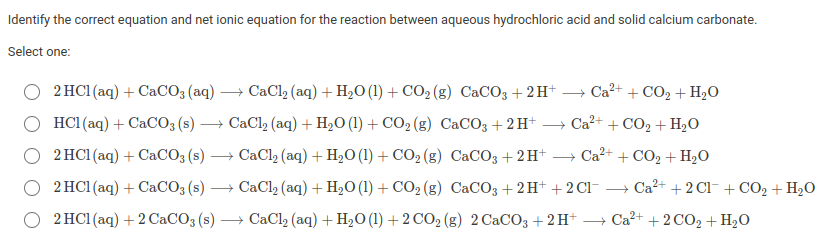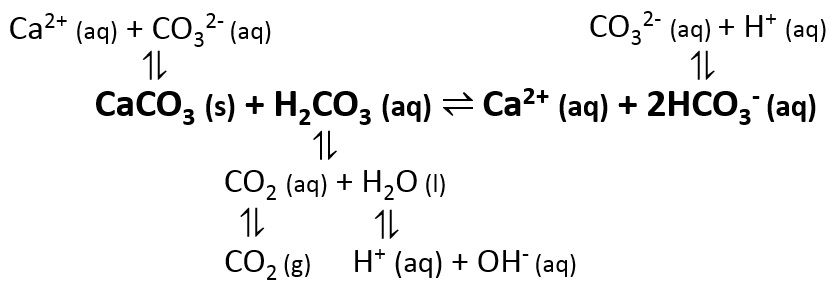35. a) Calcium carbonate reacts with aqueous HCl according to the reaction: CaCO_3(s)+2HCl(aq)→ CaCl_2(aq)+CO_2(g)+H_2O(l) . What mass of CaCO3 is required to react completely with 25ml of 0.75M HCl b) 1.0g of

Calcite solubility in aqueous solution with CO2 in equilibrium obtained... | Download Scientific Diagram

What happens when aqueous solutions of calcium chloride and of sodium carbonate are mixed? | Socratic
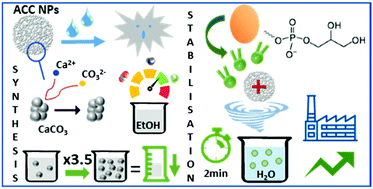
Eco-friendly processes for the synthesis of amorphous calcium carbonate nanoparticles in ethanol and their stabilisation in aqueous media - Green Chemistry (RSC Publishing)

Question Video: Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution | Nagwa

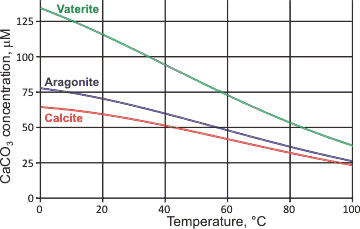
Solubility curve of different calcium carbonate forms (Warsinger et... | Download Scientific Diagram

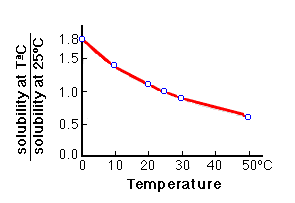
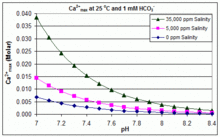
Solubility of calcium carbonate (lime scale) in water as a function of pH. | Download Scientific Diagram

What precipitate will form when aqueous solutions of sodium carbonate calcium Na_2CO_3 and calcium chloride CaCl_2 are mixed? | Socratic

The Effect of Monoethylene Glycol on Calcium Carbonate Solubility at High Temperatures | Industrial & Engineering Chemistry Research

Pharmaceutics | Free Full-Text | Ground Calcium Carbonate as a Low Cost and Biosafety Excipient for Solubility and Dissolution Improvement of Praziquantel

Solubility of calcium carbonate (lime scale) in water as a function of pH. | Download Scientific Diagram

Effect of pH and temperature on calcium carbonate precipitation by CO2 removal from iron‐rich water - Korchef - 2020 - Water and Environment Journal - Wiley Online Library

Calcium carbonate reacts with aqueous `HCl` to give `CaCl_(2)` and `CO_(2)` according to the rea... - YouTube