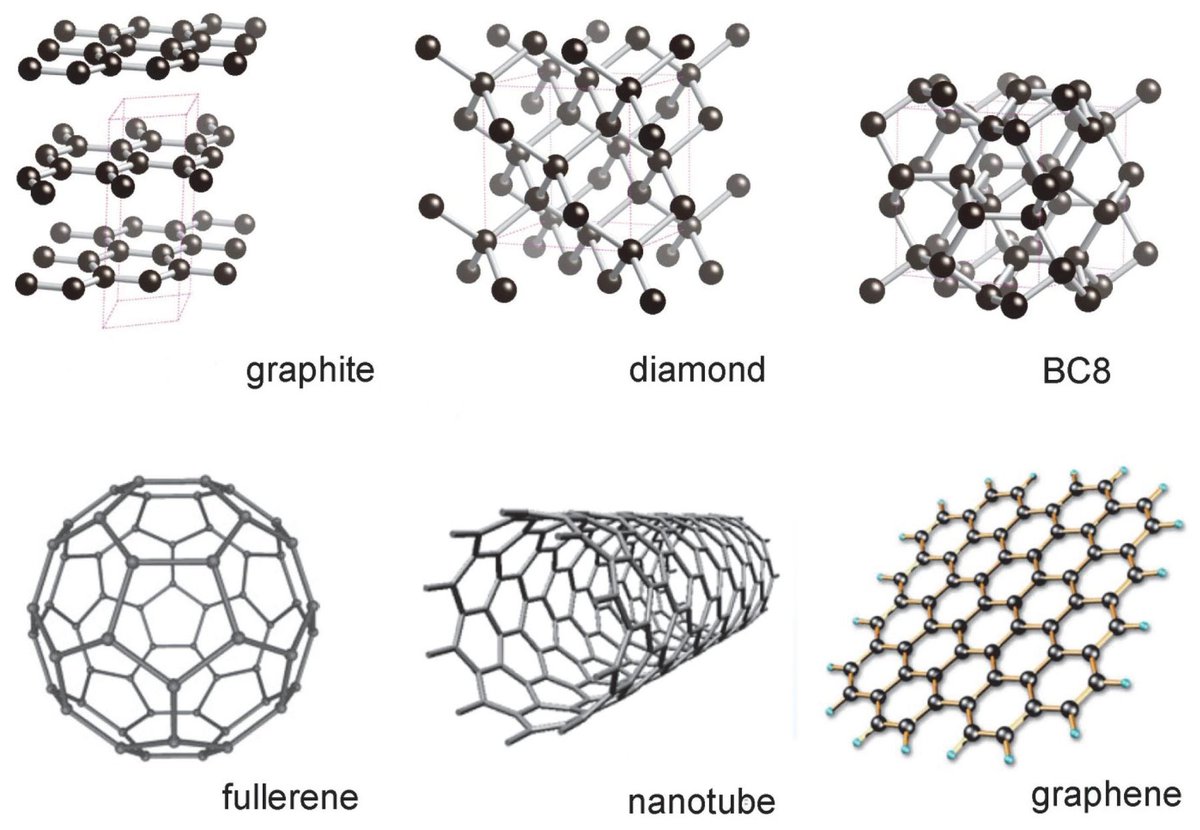
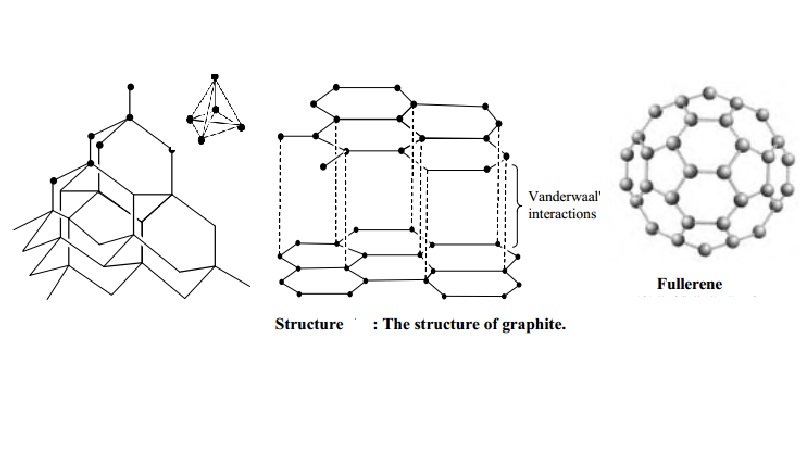
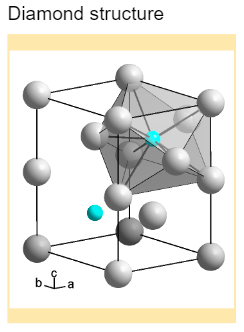
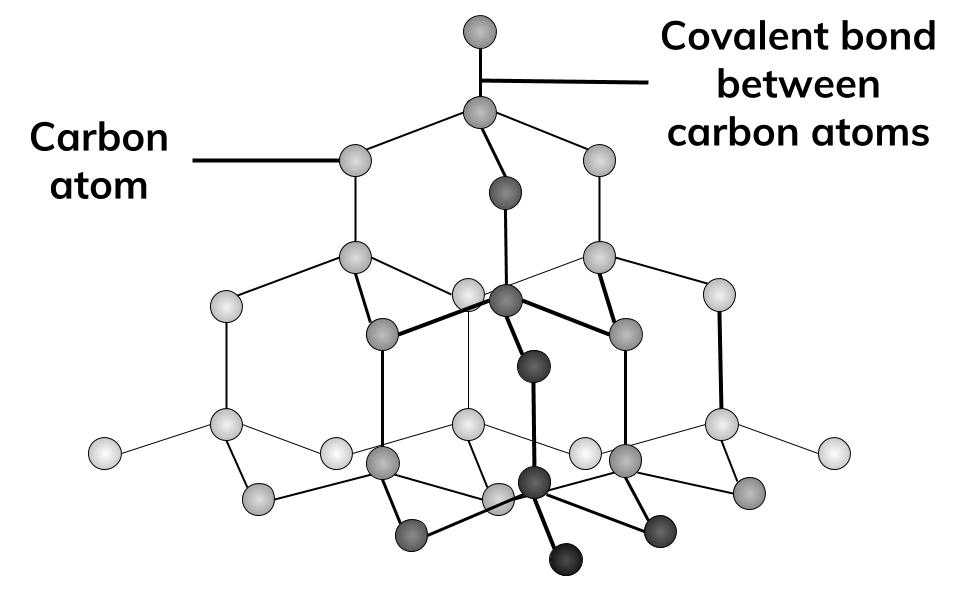
Describe the structure of diamond. Draw a simple diagram to show the arrangement of carbon atoms in it.

Molecular Structure Crystalline Carbon Diamond Allotrop Stock Illustration 1109766623 | Shutterstock
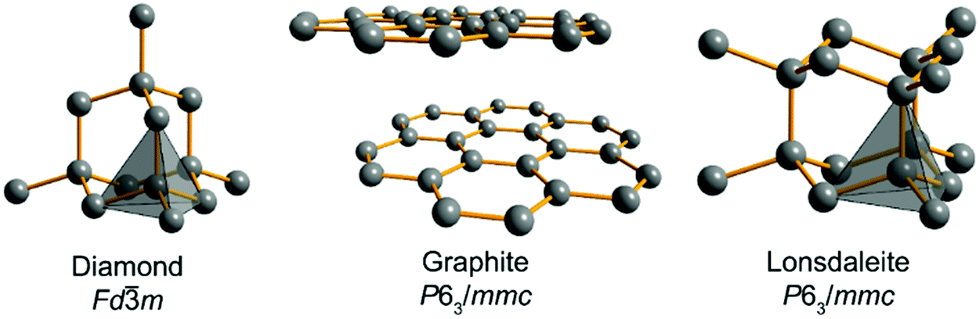
Relative stability of diamond and graphite as seen through bonds and hybridizations - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C8CP07592A

Graphite Graphite is made up of carbon atoms. Chemical formula: C It is an allotrope of carbon. Ex. Graphite vs. Diamond Graphite has the same. - ppt download
1:50 explain how the structures of diamond, graphite and C60 fullerene influence their physical properties, including electrical conductivity and hardness - TutorMyself Chemistry