Esnoderm O Ofloxacin, Ornidarole, Hydrochloride And Clobetasol Propionate Cream, Dosage Form: Ointment, Packaging Type: Tube at Rs 69/unit in Chandigarh

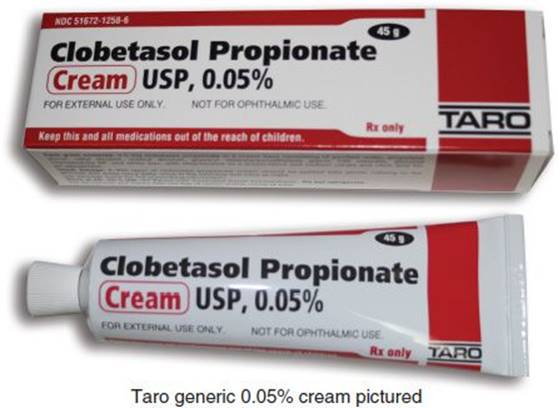
Clobetasol Propionate Cream IP 0.05% w/w at Rs 104/piece | Clobetasol Propionate Cream in Nagpur | ID: 26413772288

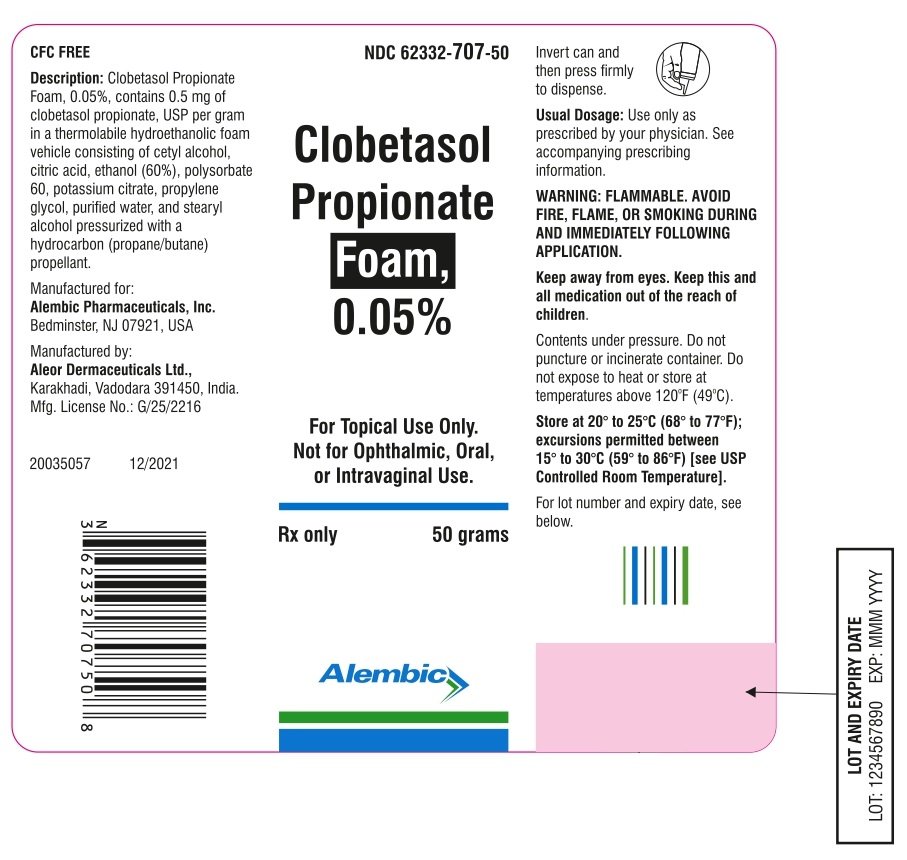
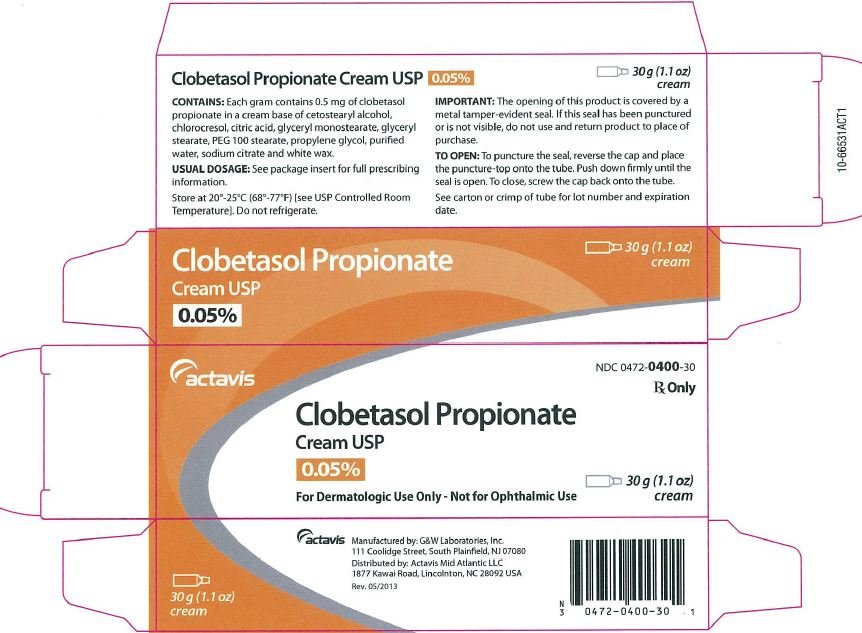
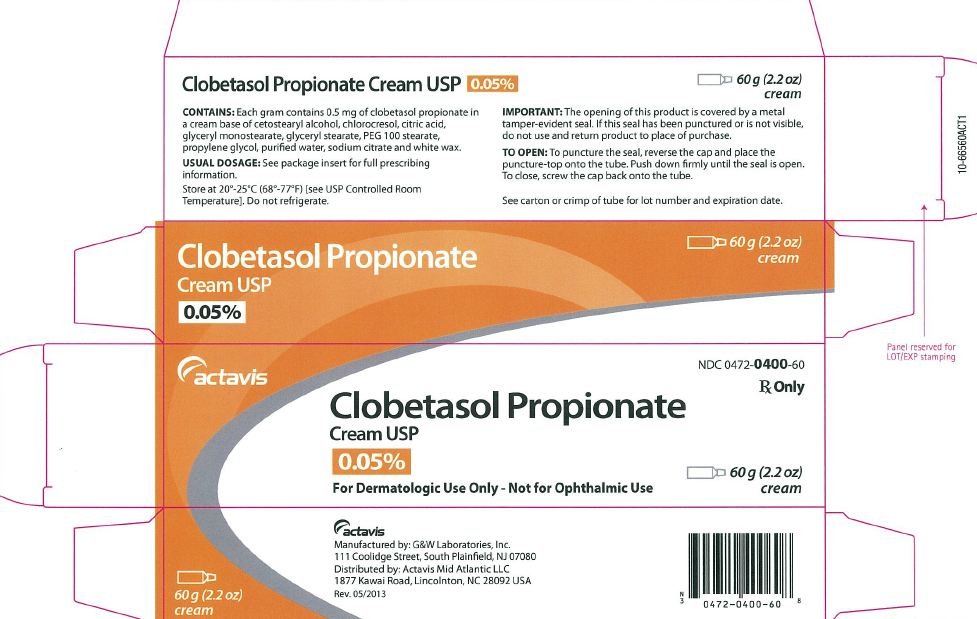
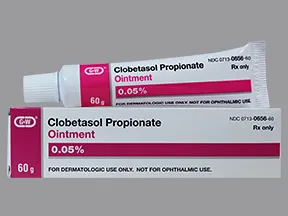
Clobetasol Propionate Ointment USP, 0.05% Rx only FOR TOPICAL DERMATOLOGIC USE ONLY - NOT FOR OPHTHALMIC, ORAL, OR INTRAVAGINAL USE.

PDF) New simple spectrophotometric estimation of clobetasol propionate in bulk and pharmaceutical dosage form

Public Alert 003/2022 – Alert on Recall of Clobetasol Propionate Ointment USP, 5%, 60g Tubes by Taro Pharmaceuticals Incorporation U.S.A, Due to Microbial Contamination – NAFDAC