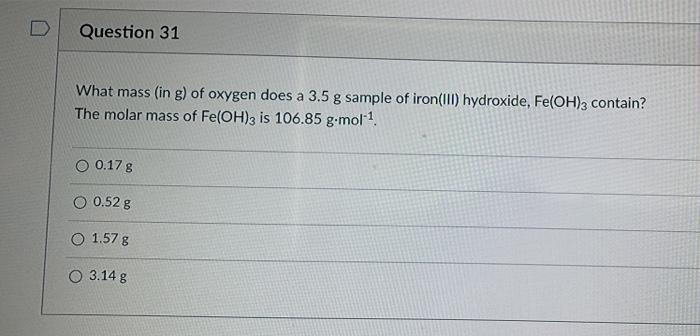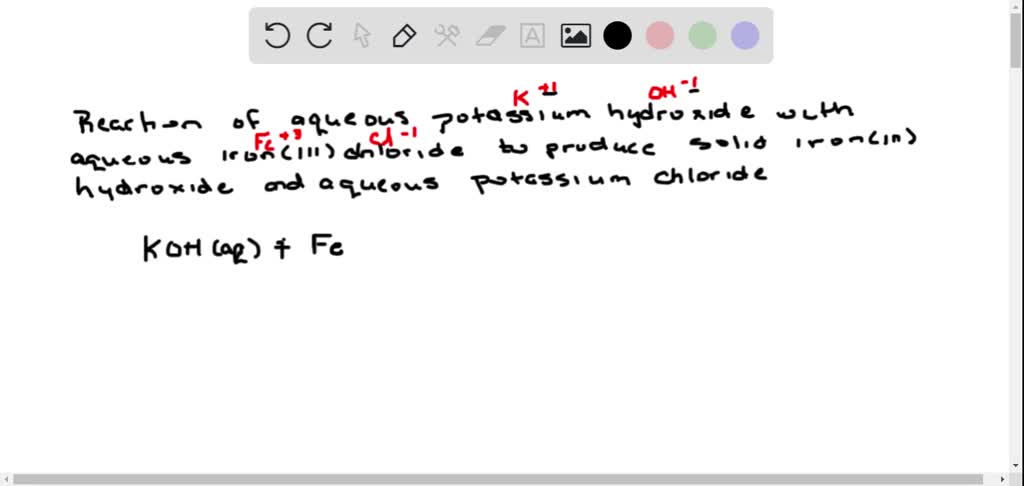
SOLVED: Iron III chloride reacts with potassium hydroxide to form potassium chloride and iron III hydroxide. Write the balance reaction, including the state of matter. Calculate the molar mass for each reactant

Molecular weight of Fe(OH)3|Molar mass Fe(OH)3|Ferric hydroxide molar mass| Fe(OH)3 Molar mass - YouTube

Molar Mass (g/mol)Table 5. Molar MassChemical NameIron (II) sulphate (ferrous sulfate)Ethyl - Brainly.ph

SOLVED: What mass of iron(III) hydroxide precipitate can be produced by reacting 75.0 mL of 0.105 M iron(III) nitrate with 125 mL of 0.150 M sodium hydroxide?
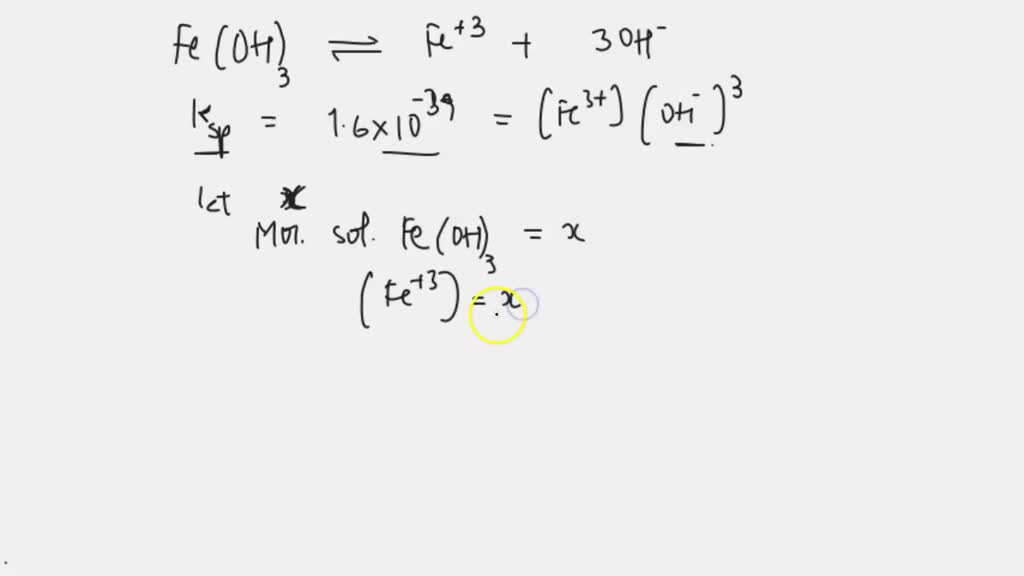
SOLVED: Calculate the Molar mass solubility of Ferric hydroxide ( Fe(OH)3 ksp for Fe(OH)3= 1.6x10^-39