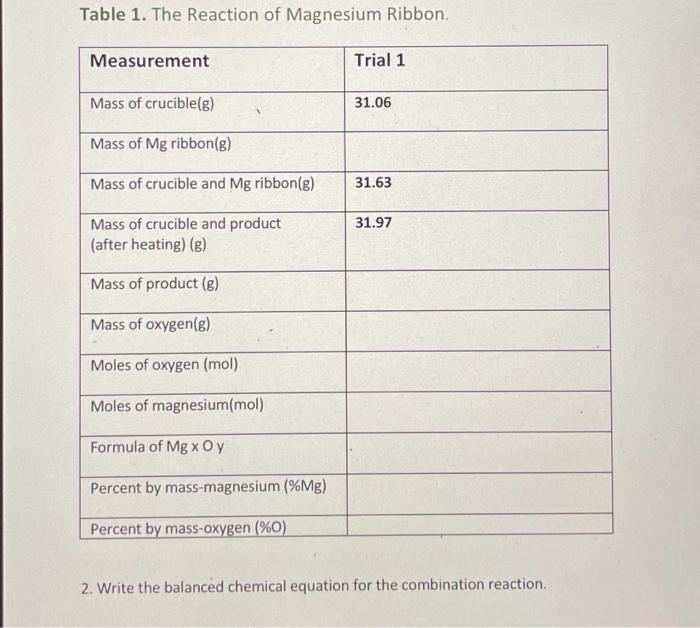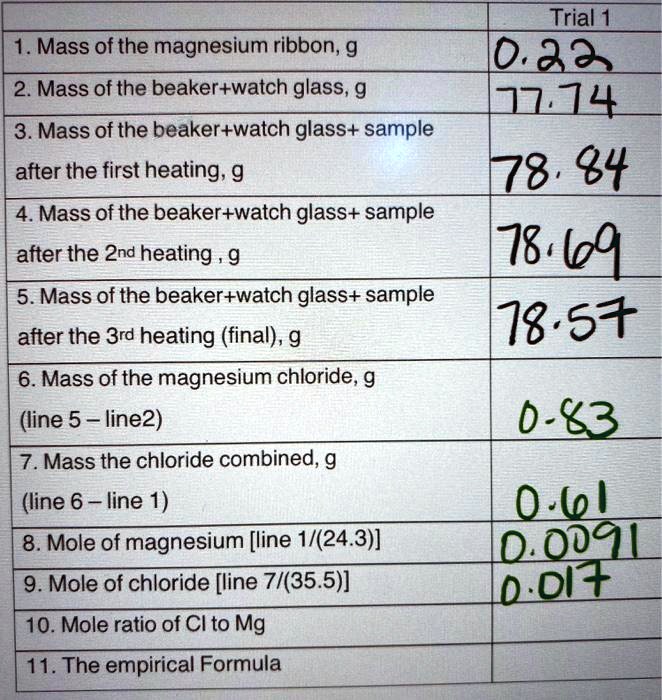
SOLVED: Trial 1 0.33 114 78.84 78 1g9 18.57 1. Mass of the magnesium ribbon, g 2 Mass of the beaker+watch glass, g 3.Mass of the beaker+watch glass+ sample after the first

PDF) Experimentally Determining of the Empirical Formula of Magnesium Oxide | Folk Narongrit - Academia.edu

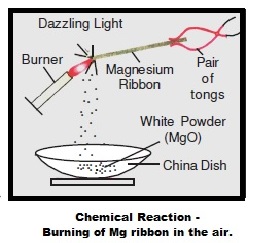
A student performs an experiment of burning magnesium ribbon in the air. A chemical reaction takes place and as a result, a white powder X forms along with a bright white light.The

A magnesium ribbon is burnt in oxygen to give a white compound 'X' accompanied by emission of light. If the burning ribbon is now placed in an atmosphere of nitrogen, it continues
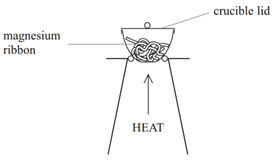
1:36 practical: know how to determine the formula of a metal oxide by combustion (e.g. magnesium oxide) or by reduction (e.g. copper(II) oxide) - TutorMyself Chemistry

Write a balanced chemical equation for the following chemical reaction : Magnesium burns in oxygen - YouTube

Magnesium ribbon burns with a dazzling flame in air (or oxygen) and changes to white substance magnesium oxide. Is magnesium being oxidised or reduced in this reaction ?

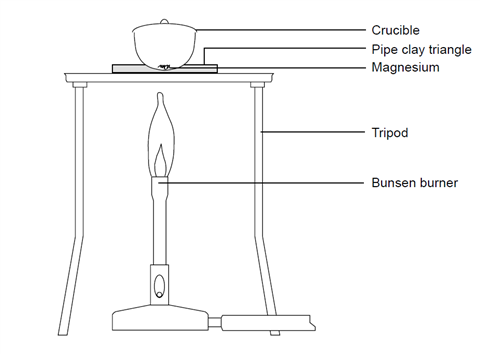
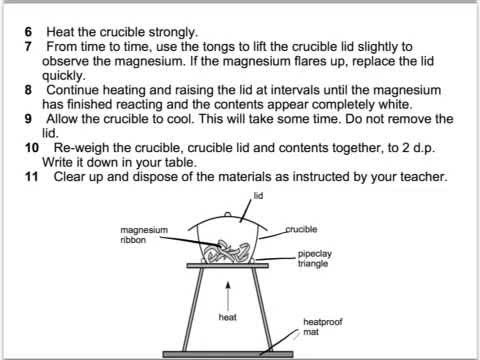
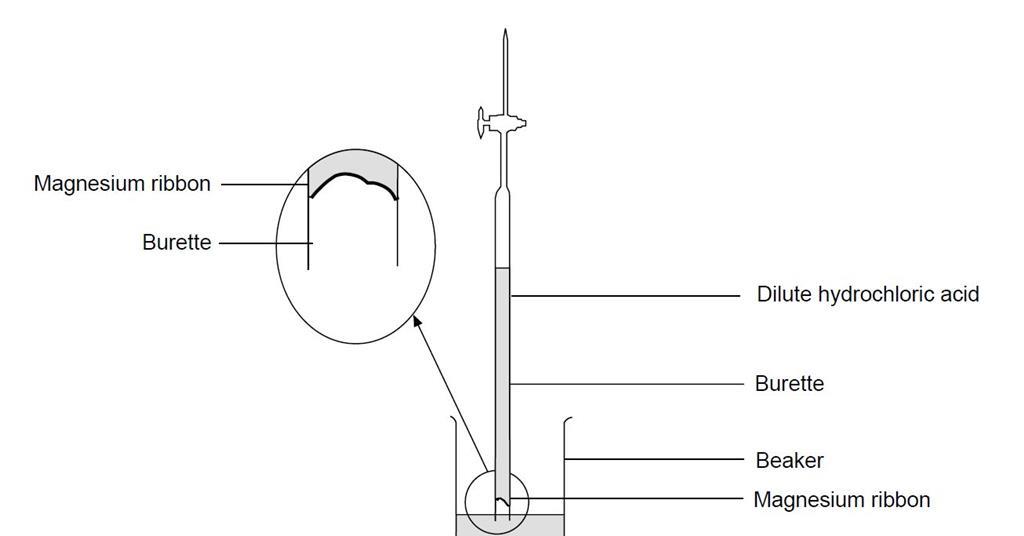
Empirical formula of magnesium oxide Background The empirical formula of a compound is the simplest value of the ratio of atoms of each element in the. - ppt download

SOLVED: Actual formula of magnesium oxide (based on charge of Mg and " 0: ZMg 2 mc (s) +02 P 275 Theoretical mass % of Mg based n the actual formula of