PDF) Long-term safety and efficacy of ozanimod in relapsing multiple sclerosis: Up to 5 years of follow-up in the DAYBREAK open-label extension trial

Frontiers | An Update for Pharmacologists on New Treatment Options for Inflammatory Bowel Disease: The Clinicians' Perspective

PDF) Ozanimod in Relapsing Multiple Sclerosis: Pooled Safety Results From the Clinical Development Program

Long-term safety and efficacy of ozanimod in relapsing multiple sclerosis: Up to 5 years of follow-up in the DAYBREAK open-label extension trial - Bruce AC Cree, Krzysztof W Selmaj, Lawrence Steinman, Giancarlo
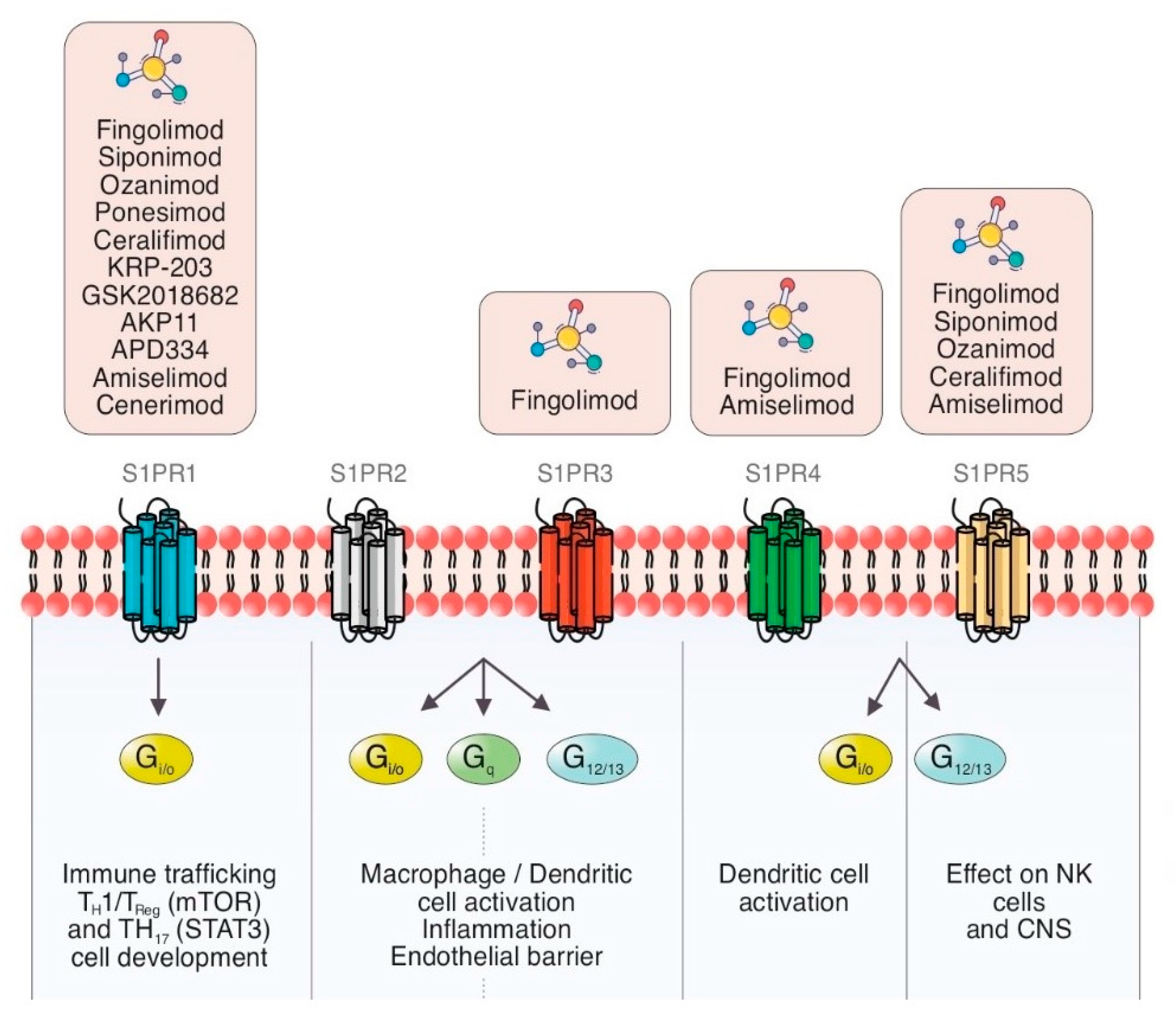
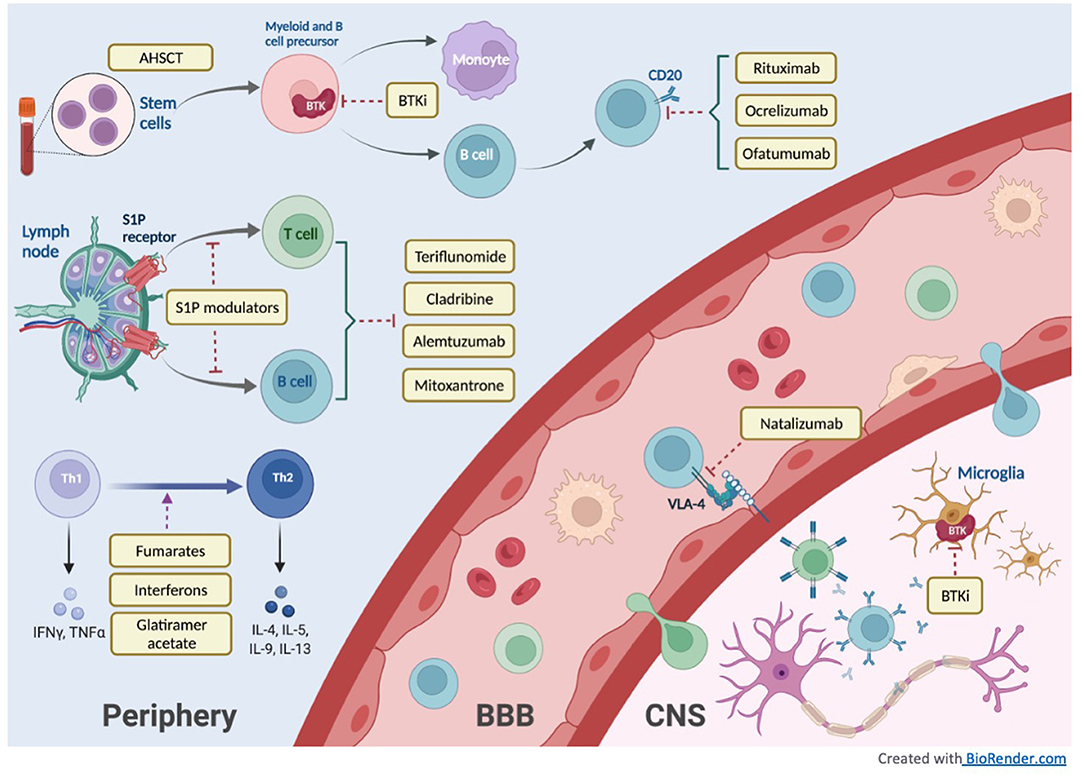
Cells | Free Full-Text | Sphingosine-1-Phosphate (S1P) and S1P Signaling Pathway Modulators, from Current Insights to Future Perspectives
Single Technology Appraisal Ozanimod for treating relapsing-remitting multiple sclerosis [ID1294] Committee Papers
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ZEPOSIA safely and effec

FDA Approves Zeposia(R) (ozanimod), for Adults with Moderately to Severely Active Ulcerative Colitis

Ozanimod in relapsing multiple sclerosis: Pooled safety results from the clinical development program - ScienceDirect

Long-term safety and efficacy of ozanimod in relapsing multiple sclerosis: Up to 5 years of follow-up in the DAYBREAK open-label extension trial - Bruce AC Cree, Krzysztof W Selmaj, Lawrence Steinman, Giancarlo

New Zeposia (ozanimod) Data Highlight COVID-19 Outcomes and Preservation of Long-Term Cognitive Function from Separate Analyses in Patients with Relapsing Forms of Multiple Sclerosis | Business Wire

PDF) An Overview of the Efficacy and Safety of Ozanimod for the Treatment of Relapsing Multiple Sclerosis

Ozanimod for Relapsed MS: Researchers Confirm Safety of Recently Approved S1P Modulator - Physician's Weekly

Long-term safety and efficacy of ozanimod in relapsing multiple sclerosis: Up to 5 years of follow-up in the DAYBREAK open-label extension trial - Bruce AC Cree, Krzysztof W Selmaj, Lawrence Steinman, Giancarlo

Peter Higgins on Twitter: "When a new drug is approved in your field, it is a good practice to go through the prescribing information in detail. There is a *lot* to unpack